When you wash old copper pennies with vinegar, the pennies react with the vinegar to form a blue compound called copper (II) acetate. Now, if you let that vinegar dry, small, black crystals will be left behind in the dish. Those are copper acetate crystals.
 It turns out you can grow much bigger copper acetate crystals. They are sleek, shiny, and look like pieces of polished obsidian.
It turns out you can grow much bigger copper acetate crystals. They are sleek, shiny, and look like pieces of polished obsidian.
Hi, I’m Chase. I’ve been growing crystals at home with household chemicals for 3 years now. From the very beginning, I’ve always wanted to grow a nice copper acetate crystal. It took me a lot of experimentation, but I finally managed to find a method to grow a gem that I’m truly proud of.
 In this guide, I’ll share what I’ve learnt, and show you how you can grow your own perfect, black copper acetate crystal at home with scrap copper and vinegar.
In this guide, I’ll share what I’ve learnt, and show you how you can grow your own perfect, black copper acetate crystal at home with scrap copper and vinegar.
Growing copper acetate crystals
Materials
To grow copper acetate crystals, you’ll need:
- 10 g of scrap copper
- A bottle of white vinegar
- 250 mL of 3% hydrogen peroxide
- Two large jars
- A big, flat container
- A small dish
- Nylon fishing line
Safety
Now, copper acetate is mildly toxic. It is not particularly dangerous when handled properly, and it’s fine to touch the crystals once in a while if you wash your hands afterwards. But avoid prolonged contact as it can cause skin irritation.
Also, it might not be a suitable experiment for young children, because you definitely don’t want them to eat it.
If you’re looking to grow safe and beautiful crystals with kids, check out my other guide on growing crystal clusters with fertilizer.
With that said, let’s get started.
Preparing the copper acetate
First, get your hands on some scrap copper. Anything that’s made of copper will do, although the most common sources are copper pipes and wires.
 If possible, cut the copper up into small pieces.
If possible, cut the copper up into small pieces.
Then, get your vinegar ready. Normal food grade white vinegar you can find at the grocery store is quite dilute – it has a concentration of 3-6%. This works fine, but the reaction will take longer.
To speed it up, you can get cleaning vinegar, which has a concentration of above 30%. This vinegar is much stronger, so be careful while using it.
 Move outdoors to carry out the reaction. You don’t want vinegar fumes to build up in your house.
Move outdoors to carry out the reaction. You don’t want vinegar fumes to build up in your house.
Add 10 g of copper metal to a non-metal container.
Next, pour in 400 mL of 5% vinegar. If your vinegar is twice as concentrated, then you should use half the amount, and so on.
After that, pour in 250 mL of 3% hydrogen peroxide. It is a common antiseptic which can be found at pharmacies or bought online.
The hydrogen peroxide’s job is to quickly oxidize the copper metal into copper oxide, which readily reacts with the vinegar to form copper acetate. Don’t use a higher concentration; a messy, brown precipitate will form.
Upon adding the peroxide, the mixture will begin to fizz as oxygen gas is released by the hydrogen peroxide. Sometimes, the fizzing can be quite intense. So, add the peroxide slowly to ensure the reaction does not become too vigorous.
 Within a few minutes, the solution should start turning blue, which is a sign that copper acetate is forming. It will steadily turn darker.
Within a few minutes, the solution should start turning blue, which is a sign that copper acetate is forming. It will steadily turn darker.
This is what my solution with added hydrogen peroxide looks like after 30 minutes.
 At this stage, you have two options:
At this stage, you have two options:
- Warm the solution on a hot plate at 70°C for 1 hour
Heating the solution will greatly speed up the reaction. However, it also releases lots of vinegar fumes, so you must do it outside or in a fume hood.
 Also, don’t carry out this step if you decide to use very strong acid – the reaction can go out of control. Make sure to take the appropriate precautions when heating acid.
Also, don’t carry out this step if you decide to use very strong acid – the reaction can go out of control. Make sure to take the appropriate precautions when heating acid.
A much safer alternative is to:
- Leave the solution outside for 2 days (concentrated vinegar) or a week (normal vinegar)
This should give plenty of time for the reaction to occur.
Regardless of what method you choose, in the end, your copper acetate solution should be a very dark blue. Now, we need to further concentrate it by evaporating away excess water.
Pour the solution into a large, flat container to evaporate outdoors, in the shade.
Wait for signs that the solution is reaching saturation. Saturation is the point where the solution contains so much copper acetate that crystals start forming.
First, you will start to notice blue-green flakes appearing on the walls of the container, just above the water level. Then, small, black specks will form on the surface of the solution. Some of it might also grow on the pieces of leftover copper at the bottom.
 Those are actually mini copper acetate crystals.
Those are actually mini copper acetate crystals.
When those specks appear, congratulations, your solution is now saturated!
You’re now ready to move on to the next stage.
If you have any problems in this stage, refer to the troubleshooting section at the end of this article.
Growing copper acetate seed crystals
From the previous step, you should have around 250 mL of saturated copper acetate solution. Of course, if you want more, just increase the corresponding quantity of copper, vinegar and hydrogen peroxide.
Filter 200 mL of the solution into a jar, close it, and leave it aside for now.
Transfer the rest of the solution into a small dish. To this same dish, add about 1 mL of vinegar.
After that, place it in a cool, undisturbed area like a storeroom or the inside of a drawer, and wait for a few days.
This will provide time for copper acetate crystals to start forming from the saturated solution. You will need those small crystals as “seeds” to grow bigger ones.
While you’re waiting, here’s a fun experiment you can do:
Place a small amount of copper acetate solution on a piece of plastic or glass and let it dry.
 Here’s what it should look like after a few hours:
Here’s what it should look like after a few hours:
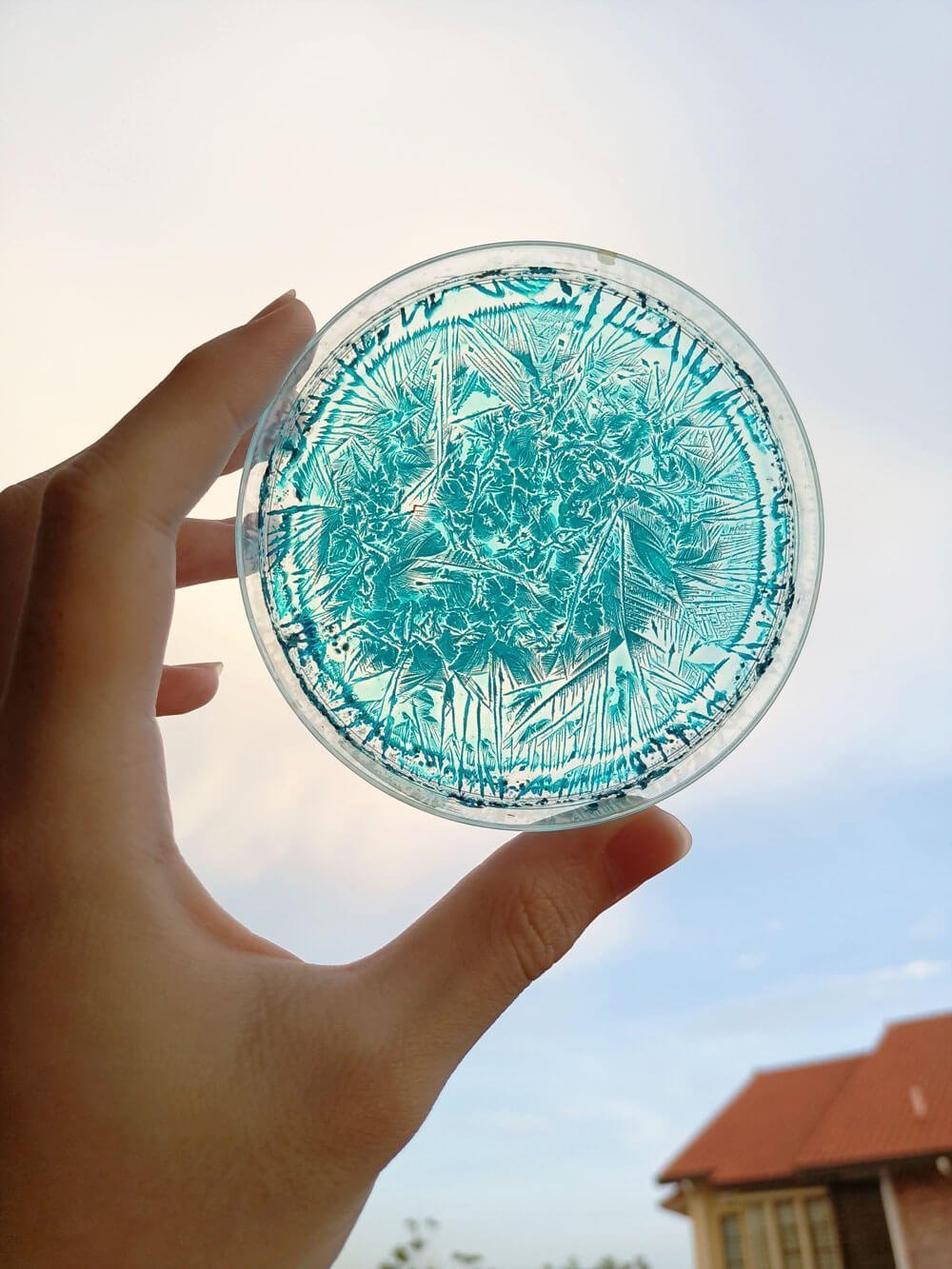 Aren’t the patterns just beautiful? Those dendrites look like rivers of blue frost creeping up a window on a cold winter’s day.
Aren’t the patterns just beautiful? Those dendrites look like rivers of blue frost creeping up a window on a cold winter’s day.
Here’s a microscopic view of those crystals, taken under polarized light by Cora A. Harris, who’s currently researching crystallography. She kindly allowed me to feature them here.
 Anyway, let’s get back to our copper acetate seed crystals.
Anyway, let’s get back to our copper acetate seed crystals.
Within the week, your dish should have formed a bunch of small, black copper acetate crystals.
 Notice how they’re much neater than the ones you saw previously. This is due to slow growth in an undisturbed environment.
Notice how they’re much neater than the ones you saw previously. This is due to slow growth in an undisturbed environment.
You can leave them inside the dish and wait for them to grow bigger, but there’s a problem.
The crystals are growing on the bottom of the container. This means that instead of being symmetrical in shape, they will be flat on the bottom. Also, left alone, the crystals will start to grow into each other.
Not good.
To fix this, first cut about 20 cm of nylon fishing line, and tie one end of it to a stick. Poke the other end of the fishing line into the copper acetate solution.
Adjust the line until it touches a nice crystal. Since there are so many, you can pick any one that is more convenient for you.
Once the line and the crystal have good contact, leave them alone for 24 hours.
 Observe the fishing line in contact with a seed crystal. They will get stuck together within 24 hours.
Observe the fishing line in contact with a seed crystal. They will get stuck together within 24 hours.
By then, the crystal should have grown into the fishing line, which means that they are now stuck together.
Good.
Now, you want to suspend the seed crystal in the 200 mL of solution that you stored in a jar earlier. Just place it like this:
 Loosely cover the top with a piece of foil / plastic wrap / paper to slow down evaporation and keep out dust. This step is very important to ensure your crystal grows nicely by reducing the risk of cracking, imperfections and parasitic crystals.
Loosely cover the top with a piece of foil / plastic wrap / paper to slow down evaporation and keep out dust. This step is very important to ensure your crystal grows nicely by reducing the risk of cracking, imperfections and parasitic crystals.
All you have to do now is to wait.
Growing a big copper acetate crystal
At the beginning, your seed crystal is still small, so it is unable to “absorb” all the excess copper acetate that wants to crystallize out of solution.
This means that many other small crystals will form on the sides of the container or on the surface of the solution.
 When they start building up, don’t bother removing them one by one. Just pour the solution into a new container, and transfer your main crystal there.
When they start building up, don’t bother removing them one by one. Just pour the solution into a new container, and transfer your main crystal there.
Some of these crystals might even decide to form on the fishing line. If they grow too close to your main seed crystal, they might merge and get stuck together.
This is no big deal if you want a crystal cluster. But if you want a single perfect crystal, you need to remove all other crystals before they grow into each other.
It’s easy to do so. Just place the unwanted crystal on the fishing line under some running tap water, and rub them off with your fingers.
As time passes, your copper acetate crystal will start growing bigger. And as this happens, you should notice that fewer unwanted crystals form.
Here’s what my crystal looks like after two months:
 The level of the solution will also start decreasing. Make sure the water level doesn’t become low enough to expose the copper acetate crystal.
The level of the solution will also start decreasing. Make sure the water level doesn’t become low enough to expose the copper acetate crystal.
If you want to continue growing the crystal, you just need to prepare more saturated copper acetate solution using the same procedure above to replenish the growing solution.
Otherwise, you can also choose to stop growing the crystal when you decide that it’s big enough. Just remove it from solution, and dry it with a piece of tissue/filter paper. Make sure you don’t wash it – copper acetate is soluble in water.
After that, you’re done! Enjoy the beauty that you’ve just created.
 Even after having grown crystals for so long, I’m still amazed at how nature creates such flat faces and crisp edges. All the crystals on this page (and on my website) formed by themselves – they were not cut or polished in any way. And yet, their form is symmetric, their faces smooth as a piece of polished glass.
Even after having grown crystals for so long, I’m still amazed at how nature creates such flat faces and crisp edges. All the crystals on this page (and on my website) formed by themselves – they were not cut or polished in any way. And yet, their form is symmetric, their faces smooth as a piece of polished glass.
Now, if you think the crystal isn’t big enough, you can of course, continue to let it grow. Unlike some other crystals like Epsom salt or copper sulfate, copper acetate crystals grow relatively slowly. But to me, their appearance more than makes up for it.
Here’s a copper acetate crystal that sat in my storeroom, growing in solution, for half a year:
 At that point, I had already refilled the solution 3 times.
At that point, I had already refilled the solution 3 times.
In the end, I decided to stop its growth after 7 months. Black and glossy, weighing in at 21.5 grams, it is undoubtedly one of the most beautiful crystals I’ve ever grown.
 And to think that such a gem was made from nothing but scrap copper and vinegar!
And to think that such a gem was made from nothing but scrap copper and vinegar!

Growing copper acetate crystal clusters
I’ve already shown you how you can use copper acetate to make beautiful patterns on glass, and how to grow a large single crystal. But you can do more with it.
It’s actually quite easy to grow crystal clusters with copper acetate. A crystal cluster is simply a bunch of crystals stuck together. To do so, just let several seed crystals stick to the string, and wait for them to grow into each other. Then, repeat the same procedure above.
Here are some amazing clusters of copper acetate crystals by Julas xZiom.
 You can even make copper acetate crystals grow on objects.
You can even make copper acetate crystals grow on objects.
For example, if you dip a piece of copper into some saturated copper acetate solution and wait for a week, some tiny black crystals will form on it. Wait longer, and the piece will be covered in big, black crystals.
 Here’s a piece of copper metal coated with small copper acetate crystals.
Here’s a piece of copper metal coated with small copper acetate crystals.
 Leave the piece of copper metal inside the solution for a few months, and the crystal cluster looks like this.
Leave the piece of copper metal inside the solution for a few months, and the crystal cluster looks like this.
You can do the same for other (non-metal) objects. Indeed, many people crystallize insects, branches and sculptures to transform them into works of art.
Storing the copper acetate crystals
Copper acetate crystals contain water molecules as part of their crystal structure. When left in open air, they can dehydrate and turn bluish white.
 A dehydrated copper acetate crystal cluster, after several months in dry air by Julas.
A dehydrated copper acetate crystal cluster, after several months in dry air by Julas.
To prevent this, you can coat them in nail polish or varnish to serve as a protective barrier. Or you can just keep them in an airtight container – I’ve kept some copper acetate crystals this way for 3 years now, and they still look black and shiny.
Troubleshooting & FAQ
-
Why is the reaction so slow?
There are many factors affecting the rate of reaction. For example, your copper pieces might be bigger than mine, your place colder than mine etc.
Generally, used and tarnished copper will react faster than fresh, shiny copper. Also, if your copper has a layer of wax/coating protecting it, use some sandpaper to sand it off first.
Regardless, don’t be worried if your solution is not as dark blue as mine after say, 1 hour, or 1 day. The important thing is that your solution is blue, which is a good sign that at least some reaction is taking place. You’ll get there, sooner or later.
-
What is this cloudy stuff that’s appearing in the solution?
 The cloudy stuff is a mixture of basic copper carbonate and hydroxide. When the copper acetate solution is not acidic enough, it can cause copper hydroxide to precipitate.
The cloudy stuff is a mixture of basic copper carbonate and hydroxide. When the copper acetate solution is not acidic enough, it can cause copper hydroxide to precipitate.
Left alone, the bluish goo will eventually cloud up your entire solution and stick to your crystals, like half dried paint.
 The matrix of black crystals embedded in greenish blue goo can be quite pretty, but sometimes, you don’t want that to appear when you’re trying to grow a big, shiny crystal.
The matrix of black crystals embedded in greenish blue goo can be quite pretty, but sometimes, you don’t want that to appear when you’re trying to grow a big, shiny crystal.
To prevent (or fix) this, just add more vinegar to the solution. It’s recommended to add more concentrated vinegar (>30%), because you’ll have to add less of it to achieve the same effect. The solution should clear within an hour.
-
Why are crystals not forming?
Copper acetate crystals only form from a saturated solution. It’s likely that your solution is not saturated enough. Continue letting it evaporate, and wait for the small, black crystals to form. They will, sooner or later.
 This is what a saturated copper acetate solution looks like. You need to wait until there’s crystalline stuff floating on the surface or black specks at the bottom of the solution.
This is what a saturated copper acetate solution looks like. You need to wait until there’s crystalline stuff floating on the surface or black specks at the bottom of the solution.
-
Will the string get stuck inside the crystal?
If you tie the crystal on the string with a knot, then yes, it will get stuck inside. But since the crystal is black, it’s barely noticeable once you cut it away.
If you poke the string into the solution to let a seed crystal grow directly on it, then you can just pull the string out after you’ve finished growing it.
-
Are these crystals fragile?
You can hold them just fine, but they are brittle and chip easily. This means that you can’t polish them. Also, you can’t wash them since they’ll dissolve in water, which makes it unsuitable for jewelry. Covering them with a coat of nail polish will help them resist wear and tear.
-
Is there another way to make copper acetate?
Yep. There are many other methods to make copper acetate. The one I mentioned above is just the most accessible for the average person. Alternatively, you can make copper acetate by:
- Reacting copper oxide with vinegar (acetic acid)
- Reacting copper hydroxide with vinegar
- Reacting copper carbonate with vinegar
- Reacting copper sulfate with baking soda to produce copper carbonate, then reacting the copper carbonate with vinegar
- Electrolyzing vinegar with a copper anode
Also, note that although old pennies (before 1982) are made of copper, newer ones mainly consist of zinc, so they will not work for the reaction.
-
What other crystals can I grow?
It’s amazing just how many different types of crystals you can grow with household materials. The easiest crystals to grow are Epsom salt, alum, and monoammonium phosphate fertilizer. All are non-toxic and beginner friendly.
If you want to do a little more chemistry, then you can also try growing potassium ferrioxalate crystals, which are emerald green.
***
That’s all for this guide. If you have any questions, feel free to leave a comment below. If you enjoyed the guide, remember to share it with your friends.
And if you manage to grow some nice crystals, do share them with me! I’d love to feature them on this website. I’m planning to create a new page showcasing crystals that others have grown.
As always, happy growing!


I was trying to do this myself and was stuck on electrolysis until I came here and realized I could just use H2O2. Whoopsie 😛
Thanks for the help!
Hi, Chase. I’m Mahiran, from India. I was looking forward to prepare some copper acetate crystals and other salts (chloride, nitrate, sulfate) as well, but I am not sure how to obtain the acids. I need some advice on that, especialy if there are any legal formalities involved.
Well the laws differ from country to country, so you’ll have to do your own research. Many of these acids are sometimes sold as household cleaning products, so you can start looking from there.
Hello,
Great work, I’ve used your guides as a reference!
I also noticed that copper acetate often tends to grow as elongated octahedra, but I’m trying to get a more “spiky” shape, closer to a regular octahedron like the one in your post:
https://www.reddit.com/r/chemistry/comments/gk6t6l/copper_acetate_i_love_growing_crystals_and_this/
Are there any additives or specific conditions that help achieve this shape, or is it mostly random?
I’ve grown both before, although I’ve never investigated this explicitly. I’m guessing that octahedra form from more acidic solutions, whereas neutral solutions prefer elongated crystals. You can try that out first.
Note that neutral/basic solutions also precipitate basic copper carbonate, so keep the solution very slightly acidic.
Hi Chase,
I have tried to make copper acetate solution by reacting copper sulphate with baking soda and then the copper bicarbonate precipitate with vinegar.But I didn’t know that colour of the solution is green not blue and there was no visible insoluble partical responsible for this colour difference. And the very small crystal is formed during evaporation of the solution ,blue colour crystal formed not black colour crystal.
Please help me to come out from this problem.
Thank you
It’s possible that the solution still contained copper sulfate, hence the crystal that formed was copper sulfate, not copper acetate. To remove the copper sulfate, you can wash the precipitate with water, drying it, and only then reacting it with vinegar.
I thought that the acetic acid (vinegar) and the hydrogen peroxide mix to become peracetic acid, not react the copper separately, or was I wrong?
The peracetic acid formed, as well as the acetic acid + hydrogen peroxide mixture both corrode the copper faster than acetic acid alone.
Hi Chase,
I am a newbie at growing crystals. I tried to grow a copper acetate crystal using a solution of copper sulfate and sodium acetate, but the crystal did not form. I think I used 1.0 copper sulfate, which might be too concentrated to grow properly with sodium acetate. By the way, do you have any suggestions for disposing of these chemical wastes? I might try to follow your steps to re-grow the crystal.
Thank you,
Richard
The other ions in the solution might be preventing the copper acetate crystals from forming. Instead of doing it this way, add baking soda to the copper sulfate solution to convert it to copper carbonate. Filter the copper carbonate, and rinse it. Then add acetic acid (vinegar) to convert the copper carbonate to pure copper acetate.
Check out this post for some advice on waste disposal.
Hi, I have quite a bit of the solution but it is particularly humid (90+%) in my city and the solution takes centuries to evaporate. I heard that alcohol can be used to dissolve copper acetate which evaporates without respect to humidity. Then, does isopropanol or ethanol grow good enough crystals , assuming I can put a lid on the container and control evaporation? Thanks
I’ve never tried that before, but feel free to give it a try!
Hi Chase. Your tutorial is really helpful. I’m planning on scaling up this whole thing, and I have a question. I’m having a hard time getting a large amount of hydrogen peroxide on my hands for a number of reasons. Is it possible to use just vinegar in this reaction? That would increase the yield while reducing the cost. I’m just not sure if the vinegar alone would dissolve as much copper as with the addition of hydrogen peroxide. Thanks in advance.
Hey Matthias. Yes, it’s definitely possible, but the process will take much longer. If you use regular vinegar, expect to soak the copper for 1-2 months before you can get a decent amount of saturated solution. Using industrial strength vinegar (caution) will cut the time in half.
Try using electrolysis to help dissolve the copper. You will want a current regulated low voltage power supply for this. Scrap copper wire will be easy to use with this technique.
Thanks for the awesome post, so excited to try this.
Just wondering, do I cover the solution for the first week while it’s outside? How does the weather affect the reaction?
No need to cover, unless the rain will get inside
Is it possible to do this with neutralized jewelry pickle? My understanding is it is sodium bisulfate contaminated with copper. I have a very blue jar of it it sitting around waiting for me to take it to hazardous waste one day, but I’m thinking now maybe I’ll try and grow a crystal? Would I need to mix it with vinegar to make it acidic again? What ph does it need to be at? The solution ends up being highly basic so not sure if it needs to be on the acidic side?
No copper acetate crystals will form, because your jar consists mainly of sodium bisulfate, which does not form nice crystals.
Even if the jar consisted primarily of copper acetate, it would need to be acidic, otherwise white sediment and basic copper carbonates will precipitate, ruining the crystallization.
What was the solution neutralised with?
Hello? I need some help
I did everything and decided to leave the solution alone for a month as my home is in a colder region than yours
It’s been 1 month now and there is no crystal formation
The solution turned dark blue like in the photo but still there is no crystal formation :-(
The fact that the solution is a dark blue means that it’s getting concentrated – it is a good sign. You just need to wait longer until the crystals form. If you really want to speed up the process, you can pour it into a flat dish to increase the rate of evaporation.
Hi, great crystals you have here! I guess one way of preserving them is to encase them in either plexiglass or some other form of non-glass translucent covering (epoxy resins etc.)
Thanks! Yep, that works.
Hi Chase,
I really want to try a copper citrate crystal grow.
Did you ever tried it?
What was the color?
Any advice?
Thanks,
Victor
Copper citrate is lighter in color, a sort of turquoise green. But I’ve not grown any crystals with it. Since copper citrate has a low solubility in water, you might have to consider growing it from an acidic solution.
The solubility of copper citrate is too low, and the solubility of the complex formed by citrate ions and copper ions is too high, both of which are not suitable for cultivating single crystals.
I’m getting a very deep blue solution but any attempt to create a seed crystal results in a bunch of very small blue crystals. The copper I’m using is the one listed in the post. Am I just not waiting long enough? Or is it evaporating Too quickly?
The humidity in my basement is 65% and I be been covering it with a coffee filter but for some reason I only get these blue crystals.
Just getting crystals is a good sign. Note that copper acetate crystals are actually blue, they only appear black when they get big enough.
It seems like there are (1) too many nucleation points in your solution and (2) the solution might be evaporating too quickly. Here’s an easy fix:
Decant the solution (without any seeds) into a new container. Then, add 2-3% of regular water to the solution (which means if you have 500 mL of solution then add 10 mL of water). This will “reset” the solution and dissolve all the tiny tiny crystals inside. Since the solution is now slightly more dilute, it will take a few more days before the crystals reform. But when they do, you should hopefully get fewer, but bigger crystals.
Awesome! I’ll try that thank you
Hi Chase, I’ve been meaning to get into growing crystals, however, I do not know what equipment to purchase. I have been seeing the burner you use, may I ask what burner you use for these? Lovely crystals btw, these is incredibly delightful!
Hello! It’s just a hot plate (without open fire) that you can place beakers on. Something like this will do.
I used pennies from before 1982 as my copper. My question is can you keep reusing the pennies or are they null?
Sure you can. After soaking it in vinegar, some of the copper might get eaten away and dissolves into the solution, but the remaining copper is exactly the same and still good to reuse.
Thank you for your comprehensive instruction on growing this crystal. My question has to do with the other methods to create copper acetate solution. Point #3 above indicated to react copper carbonate with vinegar. It seems that is a simpler way to create copper acetate solution because I can simply purchase a few pounds of copper carbonate from ebay to mix with vinegar. Have you tried that method? If yes, could you please provide some instruction on how to proportionally mix copper carbonate with vinegar to generate saturated solution? Thanks.
Yes, I’ve tried it before. The quality of the copper acetate produced depends on the quality of your carbonate. Mine was a little dirty, so the crystals that formed were not so good. Regardless, here’s a calculator for you: https://en.crystalls.info/Copper(II)_acetate
Now, add about 50% more vinegar than required on that page. This is because vinegar is volatile (which means it evaporates quickly), and also because you want to acidify your solution further to prevent copper (hydroxy)carbonates from reforming in the future.
Hey man I love this site & love to start growing crystals. I decided to start with the copper acetate crystals. I’m using the copper, vinegar & hydrogen peroxide linked here.
I originally did the procedure you detailed, 10g of copper + 250mL of hydrogen peroxide + ~55mL 45% acetic acid. After some 4 hours or so the solution turned the deep blue & was clear, I still left the solution to carry out the reaction for 2 days. I then transferred it to a flat glass rectangular bowl and left it outside to evaporate. I noticed that a white paper-like precipitate was largely forming. I added ~100mL 45% acetic acid again into soln to fix this. It worked for like an hour before it began to cloud again. I’ve left it 3 some days and about 1/3 of the volume remains and all I see is a white-teal sludge.
I tried a second run during this time. From what I understand, the overall reaction is Cu+2CH3COOH+H2O2—>Cu(CH3COO)2+2H2O right? I’ve come across this reaction online during more research.
I did a second solution using stoichiometric amounts, I calculated that in order to get 10g of desired product I would need like 3.8g of Cu. If I want to use a full 10g Cu just do 10/3.8 to find how much of the hydrogen peroxide & acetic acid I would need. I got something like 10g Cu+230mL 3% H2O2+160mL 45% CH3COOH. Not the exact numbers but something akin to that. This worked wonderfully, the soln turned light blue within like 4 minutes & in like 20 some minutes the soln turned dark blue and there was fervid bubbling off the copper bits. After 2 days I again put this into a flat, glass container and left outside. This too quickly though has started to form this cloudy white paper-like crap. I routinely went back every hour and added 100mL of 45% acetic acid until the sediment wasn’t forming again. I’ve added like 800mL of supplemental acetic acid to this soln and as it evaporates it always precipitates out this stuff :/
I decided to start filtering this stuff out and putting the filtered out liquid back into the glass containers outside, but this didn’t help at all. My first soln quickly precipitated out a thick layer of this stuff. My second soln started forming this stuff, paper-like at first & then falling down to the bottom.
I noticed the copper from the second soln (the “stoichiometric” one) had what looks like copper acetate on it. The waterlines on both glasses I have outside have a dark green/blue crystal-like line. But I can’t see any copper acetate crystals at all in either soln, not even within the milky, crappy, pale teal mush.
I have no idea what’s wrong… I’ve bought pure, uncoated copper wire & will use that in new trials, also using glacial acetic acid in lower amounts. I don’t know what’s going wrong, or what this contaminant is, or why crystals won’t precipitate out of soln even if the soln has evaporated sufficiently. This white cloudy stuff that turns to teal mush just seems to keep plaguing me.
I’ve seen others here have had the same issue but the filtering out this crap and using the remaining soln is working for me, that remaining soln is still forming the cloudy stuff. And adding more acid doesn’t work. I’ve added insane amounts of acetic acid but after evaporation it still forms the goop 🙁
I have college level chemistry knowledge, currently going to start orgo I upcoming semester so it’s not like I have no idea whatsoever, or don’t know how stoichiometry and dilutions and solutions work. But I also can’t just figure out what’s wrong. Especially without having “lab” material available. I also don’t have pH test strips but damn with the amount of acid I’ve added to soln I highly doubt it’s not acidic 😂 I don’t know what’s happening, and I can’t seem to fix it.
Any suggestions?
Thank you
Hey Gio. That’s weird. I’ve heard about others facing the same problem, but normally it goes away when more acid is added. Maybe it’s because your acetic acid evaporates away too quickly, or that it’s not the concentration that it’s supposed to be? I would try to do 2 different things:
a) Add the acid to a cloudy solution, wait till it clears up, and then completely seal the container, cutting of evaporation. If it doesn’t turn cloudy again, then it means that your problem is the acid evaporating too quickly. Of course, you can’t grow crystals this way, but at least you know what’s going on.
b) Get some pH strips to test the acidity of the solution. Compare the pH of cloudy vs non-cloudy solutions.
c) Find a way to measure the concentration of the acid, or get a batch from a different supplier. Since you’re facing this problem, you might want to look into glacial acetic acid, which is even more concentrated. That’s your best bet. Be careful though, that stuff is strong.
Thanks for answering, I have bought uncoated copper wiring & glacial acetic acid. Mixed the appropriate proportions of Cu, CH3COOH & H2O2. The solution took about an hour to get to the dark clear blue & it bubbled a lot for hours. However I came back the next day and the solution is forming this white sediment at the bottom. I did what you and others have suggested? To add more acid. I’ve been adding 100mL of the 99% acetic acid. I’ve added an incredible 400mL of supplemental acid. But now the solution is a dark blue, it’s cloudy, and almost black. I can’t be sure the acid I bought is anywhere close 99% but it is definitely strong, like more so than the 45% acid I tried first. It is incredibly potent 😂, I also haven’t added anymore H2O2.
What I did for this try is 10g Cu, 180mL of 3% H2O2 and 21mL of 99% CH3COOH. Plus the supplemental 400mL of 99% CH3COOH. The soln is now a dark teal color and cloudy, still forming some bubbles off of the copper. But I don’t see the white sediment anymore. I will try to evaporate this one after I give it another day to react and settle. I will keep you updated.
Alright, good luck!
Can thinner metal wire be used in place of fishing line? If so what the constraints on what metals the wire can be made of? Could one use ultra fine wire in the range of 20-200 microns?
Most metal wires cannot be used, because it will react with the copper acetate in a metal displacement reaction and dissolve. So the only options are silver, gold and platinum wire hahaha
You can use wire of any thickness, as long as it can hold the weight of the crystal. If you want to grow a crystal over 10 grams, you’ll need thicker wire.
About how long will it take for brand new copper to dissolve as much as yours did when you left it in there for 30 mins?
It depends less on whether the copper is used, and more on the surface area of the copper you’re using. If you use sandpaper to roughen up the new copper, and remove any protective coating that might be on it, the reaction should proceed just as fast.
I read this article 32 times before I get started to make sure everything goes according to plan; I have a limited supply of material. I come from CHINA, and I know Chinese 我是中国人呵呵。
哈哈哈加油,祝你一切顺利!
I messed it up:(
Hi Chase! I really enjoyed several of your crystal growing pages. I had looked into ADP and KTP before (electrooptic modulator and frequency doubler 1064 -> 532nm) and am amazed at the NIF process.
For copper, especially new/oily, I have been told to burn it first. Drives off oils and leaves an oxide layer on the surface to get a good start.
Thanks for making these pages!
That makes sense. I didn’t think of that. Thanks for the advice ~!
Hi chase. We accidently left our solution outside too long and it completely evaporated, but has lots of tiny little specks. What can I do with it now? Do I have to start over?
No, you don’t need to start over. Just dissolve the dried crust and little specks in some vinegar (just add enough so that everything is able to dissolve) and wait for the blue solution to evaporate again.
Thanks! My son and i had this problem too and needed an answer!
My solution WILL NOT stop becoming cloudy.
I’m using 250 mL 3% hydrogen peroxide and
And 67 mL of 30% vinegar.
I have tried adding more vinegar (a lot more) and even filtered some and put it in a jar and added baking soda to increase the pH and see if it would become cloudier and that’s not the case.
The pH is really not the issue, it’s something else. Any idea what it could be?
What did you use as your source of copper? It could be due to impurities in your copper source. Sometimes they can be quite nasty.
Was the solution cloudy immediately after you filtered it? If it was clear, how long did it take to turn cloudy again?
Not so much cloudy, but looks like bits of paper floating around. I used copper ground wire I bought from Home Depot. Which is pure copper right?
Filtering helps a bit but it turns cloudy in about 18-24 hours
Yeah it should be pure copper. I suggest you wait for seed crystals to form as usual, and then try to get one stuck to the fishing line. Then, once you suspend it in solution, the white bits floating around won’t matter much. Also, the longer you leave the solution, the more likely that all the stuff that can precipitate out will precipitate out. After a few weeks, one final filter should get rid of them for good.
You write that the gas released by the peroxide reacting with the copper is hydrogen. I’m fairly sure that it’s oxygen, from catalytic decomposition of the peroxide.
I’ve been cooking up copper acetate for some electrochemistry experiments, and the best results I’ve been getting so far have come from dissolving fine copper wire in muriatic acid (i.e. hydrochloric acid) with careful addition of tiny amounts of bleach as my oxidizer. In concentrated chloride solution, copper(II) comproportionates with copper metal to give red copper(I) chloride complexes, which the hypochlorite oxidizes back to copper(II). (Care must be taken, as excess hypochlorite combined with acid gives chlorine gas!) When the copper’s all gone, add enough oxidizer to take all of the copper in solution to +2 (bright green), then precipitate with baking soda to give copper carbonate. Wash well, then dissolve with minimal vinegar.
You’re right, the gas is more likely to be oxygen. While the reaction does produce hydrogen gas, it’s too slow to give out that much gas. I’ll have to edit my post. Thanks.
I’ve tried the electrochemical route before, and they have given great results. However, I feel that it would be too many steps for the average person to follow, hence I decided to go with the less efficient, but more straightforward method of soaking scrap copper in vinegar.
The muriatic acid / bicarbonate / vinegar process definitely takes more steps. But I got frustrated with trying to dissolve my copper with vinegar and peroxide; after a certain point, it seemed that all of the peroxide I added was just fizzing away. Using muriatic acid dissolves the copper completely, and relatively quickly. And one gets a series of pretty colours along the way: green for the copper chloride, and azure or teal as the copper carbonate precipitates.
Your crystals are very pretty. You got me thinking about a system that would suspend a growing crystal in the middle of a flowing stream of slightly-supersaturated solution, via something like the Bernouille effect. No strings or wires! But probably way more trouble than it’s worth, except as a gimmick.
Incidentally, copper acetate is only very slightly soluble in 99% isopropanol (AKA rubbing alcohol). It’s suitable for giving crystals a very quick rinse-off followed by gently patting dry, then air-drying in a warm-but-not-too-dry environment. The higher the amount of water, the more soluble the acetate is, so isopropanol concentrations less than 99% should be avoided. The acetate is also more soluble i “shorter” alcohols, i.e. methyl and ethyl alcohols, so I wouldn’t use those either.
I’ve used isopropanol to wash my copper acetate crystals before. However, one batch of them produced a brown precipitate when washed. Do you have any suspicions what might be the reason?
Hey Chase, Thank you for this amazing guide.
So, can describe how you’ve prepared copper acetate using electrolysis? I want to start growing this crystal, but due to reasons I can’t keep the h2o2 + cu + vinegar mixture outside for long.
Prepare two pieces of copper (e.g. copper pipes) to serve as the electrodes. Connect one piece to the positive terminal and another to the negative terminal. Dip them in a jar containing vinegar and turn on the power supply. The solution should start turning blue within 30 minutes. There’s your copper acetate solution. Try adjusting the current so that the reaction proceeds reasonably quickly without heating up the solution.
Thanks a lot. I’m gonna go give it a try. Also, it has to be DC power supply, ig? Correct me if AC works as well.
Yes, it has to be DC.
the electrolysis works like a charm! I just made a 1 cm crystal~ I’m hoping that it grows much bigger.
Also, I read somewhere that u could use calcium acetate in copper acetate to get crystals similar to copper sulfate. Do u know how to go about making it? I was going to go make them but then realized I had no idea what to do 😂
That’s awesome!
Yes, you can mix calcium acetate to copper acetate in a 4 to 1 molar ratio to obtain calcium copper acetate. The crystals are blue with a tint of purple, and they have a different shape compared to copper sulfate.
Dmishin’s website has some good info on how to make it: https://dmishin.github.io/crystals/calcium-copper-acetate.html
Thanks for the info about copper. My friend is interested in copper. I’ll share this info about copper with my friend.
Hey Chase, I’m trying this out for the first time and I’m in the initial stage of the reaction. I used 5% vinegar cause I am doing it with my kids. After 24 hours of evaporating, im seeing that cloudy look and a flaky sediment on the bottom of the container. It looks like I should add more vinegar to the solution according to your troubleshooting guide. Can I add more 5% vinegar or should I invest in the higher strength vinegar?
I should also mention that my copper was brand new. Should I have prepared it differently?
Hey Don. Brand new copper will react more slowly compared to scrap copper. But there’s no difference in the procedure. Sure, try adding more vinegar to the solution. If the sediment still doesn’t dissolve, filter it into a new container once the solution is saturated. If the white stuff doesn’t reappear, you’re good to go. If it reappears, consider getting higher strength vinegar. All the best!
Thanks. I have small residue building up at the edges where the solution is evaporating. Would this be evidence that my solution is saturated? Or should I wait until I see the first few seed crystals?
The residue is a good sign that the solution is approaching saturation. Wait until you see seed crystals to guarantee saturation before proceeding to the next step.
Thank you for this! I found it because I accidently made copper acetate crystals and didn’t know how or why they had formed. I had soaked very old copper US pennies in vinegar and left them alone for a few years. About a year or more ago I removed the now heavily corroded pennies but left the blue liquid in the closed mason jar. I came across it today as I was deep cleaning my studio. I was shocked to see them in there! Thank you again for all the information and explanations, I think I’ll try for the perfect big one next time 🙂
You’re welcome. I always find it interesting to hear other people’s stories about growing crystals, sometimes accidentally, just like yours!
Hi Chase,
Would you mind to confirm how much vinegar I should use if I bought the 45% concentrate you mentioned on Amazon ?
Ty
To prepare the same amount of saturated copper acetate solution I mentioned in the guide (250 mL), 50 mL of 45% vinegar should be enough. It’s best to have some extra as backup in case you want to prepare a new batch of solution.
Ok thank you. I think I was confused. I saw the 250ml, but I thought that was for 5% only and you had to reduce if you were using a stronger concentrate
So sounds like you can still use 250ml of 45% as well
Oh, actually, you should use just 50 mL of 45% vinegar *per batch*. The extra I mentioned is only for the case where you want to make *another* batch of solution.
Hi Chase,
I’ve been following your guide, but haven’t been getting seed crystals. I’ve been evaporating the solution to reach saturation, and I keep getting flakes on the the edge of the beaker and at the surface and bottom, but no crystals look like seeds, just dust. I’ve been having good luck with potassium ferricoxalate but this one is stumping me. I don’t think it’s a problem of it not being concentrated enough since flakes keep coming out of solution, and there’s no cloudyness from hydroxides.
Thanks, Thomas
Hey Thomas. It takes a surprisingly long time for already dark blue copper acetate solution to reach saturation. The flakes are a sign that your solution is very concentrated (but not necessarily saturated yet). If there are no other issues, it will just be a matter of time before tiny crystals appearing in the solution. Try to evaporate it in a dish with a larger surface area to speed things up. Good luck!
Hi Chase,
I’m very much a beginner, but I am loving this experiment. I am running into trouble though… Is this the best place to contact you??
I’ve tried twice now, after purchasing your exact items listed above on Amazon.
99.9% copper chops from Amazon
3% Hydrogen peroxide
5% white vinegar
400ml vinegar
250 mL of 3% hydrogen peroxide
and I have tried 2 teaspoons and 2 teaspoons of the copper chops
I let sit for 2 days until the solution is DARK BLUE. all seems well so far.
I place outside in a glass dish, and the solution eventually completely evaperates out and I never notice any black specs or small crystals.
What on earth could I be doing wrong? I have read through your comments and the troubleshooting guide.
Hello. I suspect the reason is because there’s actually not enough copper acetate in the solution (it might look dark blue, but there’s still a long way from saturation). I have found the rate of reaction to vary depending on the surrounding conditions. Still, it’s odd that nothing is left behind after the solution evaporated.
It’s quite hard for me to tell you what’s the problem without seeing some pictures. Do you mind sending me some pictures of your solution, and the dried dish over at chaseleanrocks(at)gmail.com?
I’m not sure why I didn’t take a picture before restarting…. I started attempt 3 tonight, so I will report back in a few days. If it doesn’t work, I’ll definitely document and send you an e-mail. Thanks so much for such a fast response! You are so awesome!
Good luck!
Hi Chase,
I started this experiment a little over a week ago. I started noticing some teeny little black specks at the bottom of the container after letting it sit in the shade over the week, so I decided to start filtering the solution today. I used a cone and some filter paper and started pouring the blue solution through, but what is filtering out is very cloudy, like the precipitate is MUCH more noticeable now. As it is filtering, it looks more and more like the deep blue solution that I was pouring into the filter (less cloudy), but I am still worried about all the precipitate in it. I was considering filtering it a few more times, but thought I would ask you first to see if it’s okay to have some precipitate left over in the solution? Not sure how this will affect my crystals going forward.
Thanks for your time and I absolutely enjoy reading your posts on crystals! I just finished chemistry and organic chemistry for the first time, so this feels like a great way to continue learning on my own! 🙂
Hello. Yes, you can try filtering a few more times. However, I suspect that the precipitate is actually copper hydroxide, which forms because the solution is insufficiently acidic. Therefore, even if you manage to filter away all of the hydroxide, it will inevitably come back. To solve it, just add a small amount (~3 mL of concentrated vinegar). If you have glacial acetic acid, it’s even better, and the solution will clear within a few hours.
Haha I’m glad you liked my posts. Trying it out yourself is definitely one of the best ways to learn!
Are the trays you use glass? Or plastic? Jus curious! Thanks for the quality how-to!
Nevermind, saw the reply to my first comment, thanks!
I noticed that you specified a non-metalic container/tray. Are the ones in your pictures glass, or plastic? Its my last obstacle before I get started! Thanks so much for the clear, well articulated guide! It’s always surprising to me how much time, research, and effort (and blood, and sweat, maybe a couple tears) go into making one even close to the caliber of yours, I really appreciate it! Hope the math degree pursuit is going well!
Thank you! The container that I used for the reaction is glass, while the dish that I used for evaporation is plastic. Actually, I have tried growing crystals using both types of containers, and there’s absolutely no difference. So don’t worry about it, both will work fine. Good luck 😀
My reaction turned red… ant advice? Only been 8 hours.
Did you use something other than white vinegar? Unwanted byproducts might form from other types of vinegar. Also, is the solution red, or is the red coming from some dust/precipitate? If it is the latter, it might just be powdered copper metal.
A copper solution will turn red if there’s lots of chloride present, and more peroxide is needed. Try adding more peroxide. If the solution instantly goes blue or green, that’s the problem. Explanation, with chemistry, follows…
The most common sources of chloride with “normal household chemicals” are salt and bleach. It’s also possible that some contaminant is dying the solution red, of course: some other metal (though I can’t think of any off-hand that are likely to be present in scrap copper that would do it) or even some kind of dye.
Copper has two common oxidation states: +1 and +2. For this copper acetate, you want (and should be getting) the +2. Under most conditions, any copper +1 that happened to be present would self-react to give copper +2 and copper metal: 2 Cu(+1) -> Cu (metal) + Cu(+2). But if there’s lots of chloride in solution, that stabilizes the +1 state and that reaction goes in the opposite direction. If that happens, you get Cu(+1), which is red, instead of Cu(+2), which is green in solutions with lots of chloride, and blue in most other water-based solutions.
Adding more hydrogen peroxide will instantly zap any Cu(+1) present to Cu(+2). The Cu(+2) will then continue reacting with any Cu that’s still around.
I see. Thank you for the explanation!
Hey, I’m not an expert in chemistry but you mentioned the copper solution will turn red if there’s too many chloride present, it is possible to make a red Cu(+1) crystal then?
This project sounds fun and I’ll try to get to it later this spring. I enjoy symmetrical shapes and natural beauty of stuff.
As a first responder, I use WISER to determine safety distances, reactions, potential hazards, etc. It is an excellent reference for those who aren’t practicing or classically trained organic chemists.
I love experimentation and learning, but know that some people don’t follow directions well.
Thanks for sharing your process and obvious passion!
This is so beautiful, thanks for sharing!
My apologies for being annoying and asking this question but can you use apple cider vinegar instead of white vinegar? I’m in the first phase of the process and it is so cool and it’s going exactly as it is supposed to however I ran out of white vinegar and I I am anxious to get to the next phase of the process!!
I can’t wait to see the crystals thank you for sharing your insight and knowledge in the subject!!
It’s better to stick with white vinegar. Apple cider vinegar will introduce impurities and organic compounds into your solution. Most of the time impurities will make stuff messier and disrupt the crystal growth. Good luck!
Thank you Chase, love your work! Looking forward to seeing more.
Working on it 🙂
Cool cool cool
Hi Chase!
Thank you for this tutorial; it is oooohsome! Question? Are their any crystals that can be grown at home then used in my jewelry? I will be melting some bismuth today !
Copper, silver and bismuth are pretty much the only practical ones that can be used in jewelry and are growable for the average person. Good luck!
You are instructing readers to mix vinegar and hydrogen peroxide together in a beaker.
The in-solution chemistry produces a toxic peracetic acid compound, which is volatile and can result in death from inhalation of vapors.
0.17 ppm in air will cause transient symptoms, 0.52 ppm in air will cause long lasting effects, and 1.3 ppm in air will cause death.
I appreciate your concern. However, the production of peracetic acid from vinegar and hydrogen peroxide is an equilibrium reaction that is catalyzed in the presence of a strong acid. This is mentioned in the Wikipedia page, and journal articles on its preparation. The most common catalyst is sulfuric acid. My procedure does not involve the addition of sulfuric acid, or any similar catalyst.
This, combined with the fact that 3% hydrogen peroxide is so dilute, an extremely small amount of peracetic acid will be produced over the course of several weeks, by which time both the peroxide and vinegar will have all reacted with the copper. If you’re still worried, remember that the reaction is performed outside, so any vapors produced will not build up.
TL;DR This method of preparing copper acetate from copper, vinegar and hydrogen peroxide is safe, and is a standard procedure.
This is a great tutorial! Cant wait to try it out, but I have a question. What is the best way to dispose of the remainding copper acetate solution?I I’ve read that it was quite toxic to aquatic animals if poured down a sink.
Yes, you’re right. I don’t dispose of my solutions, because they can be constantly reused to make new crystals. If you want to store your copper acetate temporarily, just evaporate off all the water, and the leftover dry crystals can easily be stored into a small container.
If you still want to dispose of the solution, the most straightforward way to is contact a waste disposal service. But that might be expensive / difficult to do. Alternatively, you can put some pieces of iron into the copper acetate solution, and let the iron dissolve into the solution while the copper metal precipitates out. This is called a metal displacement reaction. In the end, you will be left with iron acetate solution, which is much less toxic and can be disposed of.
Where do I leave the solution for two days?
Outside, in the shade. The exact place doesn’t matter.
This is amazing! I never knew that you could grow crystals at home! I love the clear one because it reminds me of icicles and snowflakes. I thought that it would be cool to grow them and make them into Christmas ornaments or could make decorations out of them and hang them across your window so they could reflect the light. Thank you for your article! Love*Peace*Light
dMk🦋
This is super interesting and sounds like fun, thank you for writing this all out! One question, do you think UV light would damage the crystals? I think I would try to coat them in UV resin but I worry that a concentrated UV light (6W) could potentially cause damage as it does for some other materials.
Thanks! No, I don’t think UV light will damage the crystal. But still, I can’t be sure of that under very high intensities. You can try it on a tiny copper acetate crystal and see whether there’s any effect.
Hi, thanks a lot for this guide, but I think there’s something you could/ should add:
In the beginning you dissolve 10g of copper which should react to 28.6g of copper acetate. Wikipedia lists the solubility of copper acetate in water at 20°C as 72g/l, so that’s 397ml of required water for you amount. Using 400ml of 5% vinegar that’s no problem, but if someone were to use 80ml of 25% vinegar you could run into a shortage of water and not all the copper will be able to react.
Maybe add a note about having enough water present after the line where you talk about how much vinegar is needed 🙂
Hey there. Very good observation! Actually, I accounted for that as well. The shortage of water is balanced by the fact that based on all my past experiments, not all the copper will react. This is because vinegar (acetic acid) is volatile, and some of the oxygen produced by the hydrogen peroxide is also wasted as it escapes into the atmosphere. There are ways to reduce this with equipment, of course, but I thought explaining all these technical details would make it too lengthy for the average person.
The instruction stating add 250 mL of 3% hydrogen peroxide guarantees that there will be at least a decent amount of water in the solution, so it should be fine regardless of the volume of vinegar used.
This seems really interesting and i really want to try it and was thinking of making some jewelry out of it, in this case a necklace, but before starting i just wanted to ask, is this toxic for long exposure to the skin, and if it is, is it enough to just coat them in a layer of lacquer. Amazing work by the way
A layer of lacquer will definitely help. But as a precaution, I still wouldn’t recommend it.
Hello chase, great job on growing crystals ,and sharing the knowledge of the chemistry required to make them. I have always been fascinated with mother nature’s crystals being a rock hound in my younger years. And as a adult, I am curious about the use of crystals in the solar energy field. Your use of copper is what grabbed my attention. Do these crystals have any electrical properties? To make a homemade solar PV cell copper is heated with flame or fire, and the black specs that are formed are semiconductors and the first to be used to turn sunlight into electricity. Not very efficient but it works. Silicone crystals can be grown in a gravity free environment to the size of a basketball. Anyway what I’m leading up to is, can a crystals from a cannabis plant be grown to take advantage of their light refacting properties to make solar panels more efficient ?
Hello there. I know that copper acetate crystals are used in neutron spectrometers. Other crystals such as MAP and Rochelle salt have piezoelectric properties while sodium chloride crystals are used in lasers. However, I’m not knowledgeable in renewable energy or electricity, and I have never grown CBD crystals, so I’m sorry, I have no idea.
Chase, email me sometime. I can elaborate on my experience which I never expected. I started simply cleaning fossils and ended up with odd and amazing crystals.
Jeff
cdsjdsmjs@aol.com
You got me interested. Emailing you right away!
Last year I was cleaning fossils in 30% vinegar and placed random interesting rocks into jars and covered them with extra vinegar to see how they would clean up. Each jar proceeded to form amazing and different crystals on the lip of each jar and then covered the rocks themselves as the liquid evaporated. I still have the crystals and amazing photos for anyone interested. Jeff in Ohio at cdsjdsmjs@aol.com
I’d love to see photos of your stuff! Thanks!
Terry Sisk
Illinois, USA
interested in creating custom minerals for my jewelry. Can I anchor the chrystal to something other than fishing line filament? What other materials can be used without upsetting the mineral balance?
What other materials do you have in mind? You can grow them on a thread, a string, a branch, rocks, seashells, dead insects, skeleton leaves, ceramic, even concrete. Most metals do not work, because they will react with the copper acetate solution. Copper, silver, gold and platinum are the exceptions.
I just started the process for making these today and I’m super excited to see what happens! Thank you so much for all the instructions.
You’re welcome! Feel free to ask me if you have any questions during the process.
Very cool. I’m ready to give this a try. A couple of questions:
1) Is there any reason not to use 325-mesh copper powder? It’s avsilable for about 15% cheaper than the shavings. I realize the initial reaction will go faster.
2) Would there be any advantage to more concentrated H2O2? I have 35% that I can dilute down as needed. Obviously it would liberate H2 more
vigorously. Thanks much!
1) You’re right. I had used pieces of copper metal all along, so I didn’t think of the powder version. Thanks ~ I’ll edit the post!
2) You can certainly use more concentrated H2O2. But don’t pour solutions of above 30% directly to the vinegar and copper mixture. It will cause a lot of insoluble brown stuff to form. I’m not sure what that is, I’ve heard some people say it’s copper peroxide. Regardless, it’s messy and very hard to get rid of.
Note that if you use both copper powder and a stronger peroxide, the reaction will be very vigorous. Be careful, and add small amounts at a time.
I love your crystal-making library! Do you research about how to make each type of crystal before you try making them or do you use trial and error to figure out techniques and the optimal quantities of solutions to use? Your ideas are truly inspiring for people to discover some cool ways of using chemistry! If I had known about this back when I was in university or AP chem lab, I would have been hooked on it as well. You have a great way of presenting things and I hope you will continue to teach everyone about crystal making chemistry! I hope you plan to teach chemistry at some level in the future. You’d be excellent at it!
Thank you very much 😀 I do some preliminary reading online, and use other people’s experiences as reference (most of them come from r/crystalgrowing). Then I get my hands on the chemical and try to grow a crystal using the standard procedure. I then tweak the variables to suit the equipment and weather conditions I have.
Ikr, it’s such a cool way of getting people into chemistry. In fact, I first started growing crystals because of my high school teacher. Thank you for your kind words; I’ll continue to share my hobby here. Actually, I’m currently taking a degree in mathematics, and I hope to teach math one day haha
Rather than use vinegar would Citric Acid work?
This is great information. I will use your crystal growing to teach my grand kids about chemistry. And have some fun of my own too.
No, citric acid will not work, because you will make copper citrate instead of copper acetate, and copper citrate does not form nice crystals.
BUT the good news is, you can crystallize citric acid by itself! Citric acid crystals are non-toxic, are transparent and grow quickly.
Regarding pennies as a copper source, they have been almost all zinc, not copper, since 1982. Today’s pennies are 97.5% zinc and only 2.5% copper. I wonder if that might grow nice zinc acetate crystals?
Ah my bad. I should edit the post, thanks for pointing it out. Zinc acetate indeed forms crystals, but they aren’t quite as nice as copper acetate. You can see some photos on the Crystal Growing Wiki: https://en.crystalls.info/Zinc_acetate
Hi Chase, I’m a 69 year old retired man in Tasmania. Your scientific enthusiasm is infectious for laypersons also.
I do coppersmithing using recycled cu plumbing pipe off-cuts, beat annealed sheet copper into bowls and solder flared pipe onto the bowl with lead/tin solder and make a variety of handles hammered twisted from scrap pipe. Handles are rivetted on using cu leather rivets and washers.
All the best in your studies and future career.
Kind regards,
Charl.
I’m happy to hear that, and thank you for sharing. I know it’s currently not possible, but I just want to let you know that I would be very glad to have a tour of your workshop. Much respect.
This sounds awesome, can’t wait to try it out!!!
Wow this is AMAZING!! I had no idea such a thing was possible!!! I will be giving this a try. What do you do with them?
Good luck! I keep them in my collection (I’m a big mineral fan), show them to my friends, give some of them as presents. I’m currently trying to persuade a few of my friends to start growing crystals, and big shiny specimens definitely help. I suppose I might be to sell a few crystals one day, but I’ll have to sort out the details like preservation and shipping first.
Not only is the Chemistry fun and the break-down well articulated but this site is lovely. I’m a former Chemistry major who ended up leaving school before finishing, I’m currently in a User Exeperience Design class for a transition into the tech industry and this site is a perfect blend of my two passions 🙂
Keep up the great work, I’m definitely going to follow some of your guides once I’m done with this class and have more attention to spend elsewhere.
I am so thankful to exist at the same time as technology that allows for such vivid sharing of passions. Thank you for taking the time to share this! This is what I love most about humanity.
Thank you so much. I really love this hobby, and I’m glad you liked it. Have a nice day!
These are great and very beautiful. Are these crystals safe to handle without a glove afterwards? If we cover it in enough varnish will they be able to last? And can we use pennies as the starter copper? Or if I have a copper cup can I just pour the vinegar in that?
Thank you! Yes, you can hold them without a glove, just wash your hands afterwards. It will only cause skin irritation if you hold it for a very long time. Yep, varnish will definitely protect them, though I still wouldn’t put the crystal in water. And yes, pennies will work. The copper cup is not a good idea. Over time, the walls might become so thin that the solution will leak out and cause a mess.
I am a huge crystal fan. I live in western nc and hunt for crystals in my back yard and creek area. We find tons and tons of beautiful crystals and clusters. Its a great hobby to get into especially if you want get away and enjoy yhe peace and quiet. I am super stoked about this project seeing that we only find clear and white crystals. I am always looking down on the ground for crystals in gravel driveways and yards i am so addicted. They are so beautiful. I am still interested in knowing how these crystals come about in the ground, why they are found in our area and what the story is behind them. What’s their story and how old are they? So far, i’ve come up with no rhyme or reason. If you could shed light on this, it would be fantastic. I also find indian artifacts like pottery and arrowheads but my mind goes to the beautiful crystals. I find myself rummaging for hours and hours on end hunting for them. They are gorgeous and breathtaking. Nature is a mysterious and rewarding thing. Thanks for educating us on making ones at home that are different colors and very cool looking. A great conversation piece for sure.
Hey Amy. When I was young, I was always looking everywhere on the ground for crystals too! I didn’t get any impressive specimens, but I did manage to find some small quartz crystals at parks, hiking trails and so on. One time I found a small transparent one, and I got so excited I kept looking at it for weeks! Your comment made me relive all those childhood memories again :))
Those crystals naturally form deep in the ground under high temperature and pressure. Over time, minerals in the earth’s crust solidify and crystallize into these beautiful shapes. The presence of elements such as iron and copper give some crystals their color (iron is green, copper is blue). Then, these crystals are brought to the surface by geological activity. This same activity causes fossils of prehistoric animals which once lived in ancient seas to be found on the Himalayas.