Potassium ferrioxalate produces striking green crystals that are both beautiful and easy to grow.

Potassium ferrioxalate is a chemical that can be made from relatively common materials. You can prepare it by dissolving rust in oxalic acid, which is often used in cleaning, and then neutralizing the brown solution with potash (potassium carbonate).
The resultant solution will look bright green, and when it evaporates, crystals start to grow.

Hi, I’m Chase, and I love to grow crystals at home. Not many people know that it’s easy to grow crystals with household chemicals like table salt, Epsom salt and sugar. I first started this hobby in high school and I’ve been loving it ever since. Today, I’ll share how I grew potassium ferrioxalate crystals with you.
If you want to try it out, note that while the compound is not particularly toxic, it’s still an irritant. Also, making it involves dissolving rust in acid, so wear gloves during this step.
If you’re looking for a fun activity to do with your kids, making green rupees probably isn’t the best choice. Consider checking out my guide on how to grow alum crystals, which are both beautiful and food safe.
Now, let’s get started.
As mentioned, you need rust, oxalic acid and potassium carbonate to make this compound.
I made the rust myself, and bought the other two online. Oxalic acid is commonly used in cleaning and bleaching. Potassium carbonate is sometimes used to make certain foods. It also acts as a drying agent.
How to make rust?

Rust is iron(III) oxide. It can actually be bought as well, but I thought it’d be fun to make it myself.
I first soaked some fine steel wool in a dish containing salt water. Salt solution speeds up the rusting process, just like how objects rust faster near the sea.

However, it was still going to take a long time. Although I only needed about 5-10 g of rust to grow decent crystals, it would take weeks – even months for the piece of steel wool to crumble completely into rust.
To further speed things up, I added some hydrogen peroxide to the steel wool. Hydrogen peroxide is an oxidizer, which helps convert the iron to iron(III) hydroxide.
 The next day. I added hydrogen peroxide only to the dish on the left.
The next day. I added hydrogen peroxide only to the dish on the left.
I continued to periodically add hydrogen peroxide to the dish on the left. After 2 weeks, the treated steel wool had completely disappeared, while the untreated one wasn’t even halfway there.

I filtered off the salt water from the dish on the left, and washed the iron hydroxide a few times to remove traces of salt.
 The iron hydroxide looks almost exactly like the floor. Oh well.
The iron hydroxide looks almost exactly like the floor. Oh well.
Then, I heated the iron hydroxide strongly at 200°C for 1 hour to convert it into iron oxide. I generally heat it for twice as long as the time it takes for it to dry completely to ensure all of the iron hydroxide has reacted.
Upon heating, the color turned from black to reddish brown. Pure iron oxide is actually red, but small amounts of impurities aren’t a big issue for crystal growing.

The initial piece of steel wool weighed roughly 1 gram. I ended up with 4 grams of dry iron (III) oxide powder, so the yield was good.
A more efficient way of making rust is via electrolysis. The disadvantage is that it requires a power supply.

To carry out electrolysis, I first prepared dilute salt solution as the electrolyte. Then, I used a piece of iron as the positive electrode and a steel wire as the negative electrode.
Once I turned on the power supply, the iron started dissolving slowly, while bubbles of hydrogen gas formed on the wire. Hydrogen gas is explosive, so I did it outside. Some of you might have concerns that chlorine gas will develop, but because chlorine is so reactive, and because the salt solution is dilute, it’s safe for this setup.
A brownish precipitate soon formed at the bottom of the container. After half a day, I filtered and dried it, just like the steps above, yielding 6 grams of rust.
This video by NurdRage explains the process in more detail.
Enough about rust. This article isn’t titled How to Make Rust.
We’re here to grow shiny green potassium ferrioxalate crystals.
Making the potassium ferrioxalate

To prepare the solution, I dissolved 45 g oxalic acid in 200 ml of warm water. Then, I added 6 g of iron oxide, stirring as the solution slowly turned reddish brown.
Note: If you’re using anhydrous oxalic acid, just 30 g is enough. It’s usually sold as the dihydrate version, which requires 45 g.

It took a while for the rust to dissolve. I continued to heat the solution gently on the hot plate for a few hours, until most of the solid stuff had dissolved. By then, the solution, iron(III) oxalate, was dark brown.

I then added potassium carbonate into the solution, bit by bit. There was a lot of fizzing as the reaction produced carbon dioxide. In total, I added 15 grams of potassium carbonate, and eventually, the solution turned greenish.
I stirred it around a bit, turned off the heat and waited for the precipitate to settle.

Note that at this point, the solution was supposed to be a striking fluorescent green, but it was a dirty olive green instead. Based on my previous runs, this could be due to 2 reasons:
- Not enough potassium carbonate was added
In this case, there’s a lot of brown iron oxalate solution left over, so naturally the solution wouldn’t look very green.
- Not enough oxalic acid was added
Provided there’s enough potassium carbonate, it seems like a small amount of excess acid makes the solution more stable. Basic solutions quickly turn brown and form precipitate. After adding a little oxalic acid, they immediately revert to green.
 Solution with extra acid (left) vs solution without extra acid (right).
Solution with extra acid (left) vs solution without extra acid (right).
So I just added 2-3 g more acid, stirred it around, and filtered the glorious green solution into a dish. Then, I placed the dish inside the storeroom to provide a sheltered, stable condition for the crystals to grow.

Growing potassium ferrioxalate crystals
As water evaporates, the solution slowly becomes more concentrated. Once it reaches saturation, the extra solute has nowhere to go, and it crystallizes out.
My solution was a little dilute – I guess I should have heated more water away at the start to speed up the crystallization. Regardless, after 5 days, tiny green crystals started forming on the surface.

They eventually dropped down and continued growing at the bottom. Here is what they looked like after a week:

The crystals aren’t very big, but gosh, they’re so pretty!
Also note that the solution became much greener than before. I’ll explain more about this later.
By the 11th day, I had three green rupees sitting nicely in the solution, while many smaller crystals formed at the side. To prevent them from growing into each other, I blew them apart using a plastic dropper.

Observe that the crystals have straight edges and flat faces without being cut or polished. This is the natural crystal structure of potassium ferrioxalate. Meanwhile, other crystals such as table salt grow into cubes, while alum forms crystals shaped like octahedrons.

By day 17, the crystals were getting quite large, and mold had started to form in the solution. This is because some bacteria are able to use oxalate as a food source. I didn’t appreciate that though, and I wanted to try out a different technique, so I decided to remove them from solution.
First, I poured the solution into a different container. Then, using tweezers, I picked them out and placed them on a piece of filter paper to dry.
And the results were breathtaking.




Amazing!
But I wasn’t done yet.
I wanted to grow bigger crystals.
Growing big potassium ferrioxalate crystals
A common crystal growing technique is to tie a seed crystal to a string, and hang it from solution. This way, other crystals won’t stick to it, and it will also grow more symmetrically.
I picked a small, but beautiful crystal from the batch I had grown earlier, and tied it to the end of a fishing line. Then, I taped the other end of the fishing line to a stick, and lowered the crystal into a cup containing the potassium ferrioxalate solution.


I also covered the top of the cup with some cling wrap to slow down evaporation and discourage extra crystals from forming.
The crystal immediately began to grow, and after a few days, a nice clear layer had formed around the original crystal.
 You can still see the outline of the original crystal.
You can still see the outline of the original crystal.
Meanwhile, lots of smaller crystals also started forming at the bottom of the cup. These would compete with the main crystal and slow down its growth, so I transferred it to a new container.
With fewer crystals acting as competition, it began to grow much faster. Also, the sides widened out nicely. By the 1-week mark, it looked like this:

Unfortunately, some temperature changes due to the weather caused small cracks to form inside the crystal. They’re only visible when I shine at it with a torch.
Also, as mentioned previously, mold likes to form on the surface of the solution. Although they didn’t really affect the crystal growth, I removed them periodically with tweezers.

I also tried scooping them up with copper wire, as copper is toxic to microorganisms. It worked for a few days, but the mold always came back eventually.
After 3 weeks, the crystal was getting big, and the level of the solution had greatly decreased. I decided it was time to harvest the crystal.


I removed the crystal from solution and dried it using a piece of filter paper. Then, I cut the string away using some scissors. The final crystal was 4.5 cm long and 2.6 cm wide.
It was undoubtedly one of the most beautiful crystals I had ever grown. The faces of the hexagonal crystal were sharp and crisp, and it glowed fluorescent green under the morning light.



Storing the crystals
The deep green color of potassium ferrioxalate is breathtaking. However, like all ferrioxalates, it is sensitive to light. This means it decomposes in the presence of bright light, with a reduction in the iron center and the oxalate ion oxidizing into carbon dioxide.
Green potassium ferrioxalate solution exposed to light will turn yellow, and then brown. After keeping them in the dark for some time, they become green again as the iron ions are oxidized by air, and the ferrioxalate complex reforms. Adding small amounts of oxalic acid greatly helps to speed up the process.
This explains why my initial solution was a little yellowish, and why it turned bright green after sitting in the storeroom for a few days.
 The solution on the left was keep indoors, while the solution on the right was exposed to direct sunlight for 30 minutes.
The solution on the left was keep indoors, while the solution on the right was exposed to direct sunlight for 30 minutes.
On the other hand, the surface of crystals exposed to bright light will turn duller, and eventually white. I have found that occasional indoor lighting is fine. But this process becomes much faster under the sun.
Here’s the first batch of crystals that I grew, and brought outdoors for 30 minutes, compared to those that I did not take outside:
 Actually, they don’t look that bad – there’s a different vibe to them.
Actually, they don’t look that bad – there’s a different vibe to them.
When kept in the dark, these crystals are quite stable. I have kept them for a month or so, and they look the same.
How impurities affect crystal shape
You can also change the shape of potassium ferrioxalate crystals by adjusting the acidity, and ratio of potassium carbonate to iron (III) oxalate. Impurities also affect its structure, ranging from sticks to hexagonal crystals.
 For example, this beautiful crystal by Reddit user u/cwdcloud has a different shape from mine.
For example, this beautiful crystal by Reddit user u/cwdcloud has a different shape from mine.
You can also find pictures of much longer, stick-shaped crystals online.
I have seen small octahedral crystals before, growing in a solution with a large excess of oxalic acid (I added too much by accident).
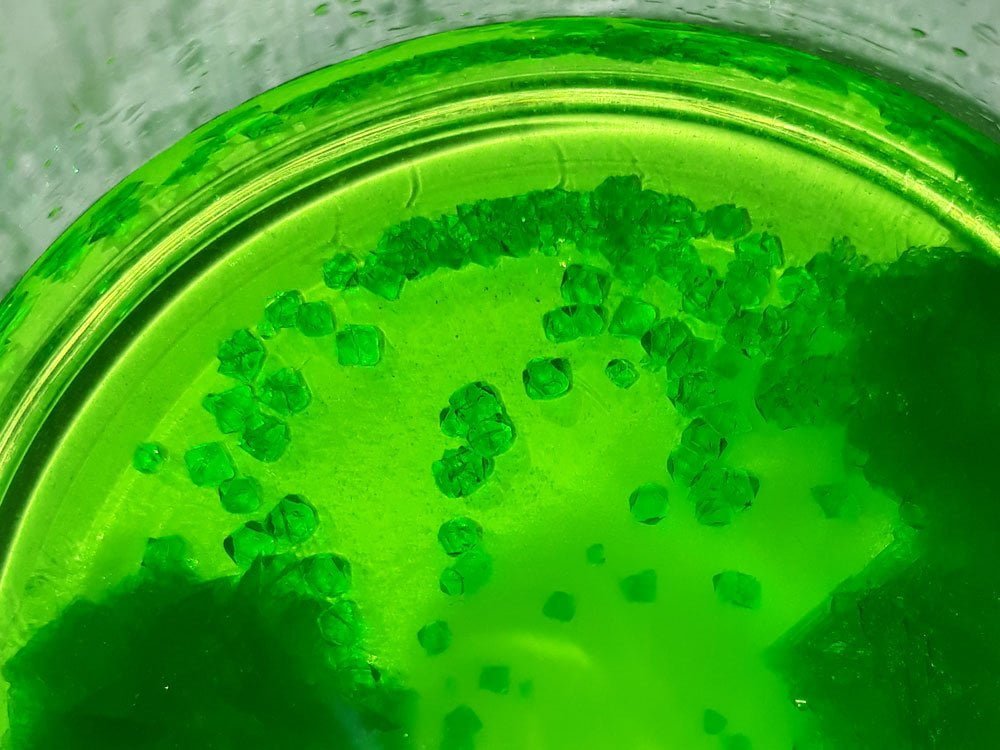 You can see the octahedrons, as well as some clusters of oxalic acid crystals intertwined with potassium ferrioxalate.
You can see the octahedrons, as well as some clusters of oxalic acid crystals intertwined with potassium ferrioxalate.
I have yet to pin down the exact conditions affecting its growth – I’ll have to make more experiments. Crystal growing is both a science and an art, and the results amaze me every time.
Edit: I’ve figured out the effect of excess acid on the shape of the crystal. To a saturated solution of potassium ferrioxalate, I added oxalic acid until the solution was also saturated in terms of oxalic acid. Then, I used the same procedure as above. Here are the results:


They aren’t actually octahedrons. Compare it to the rupee shaped ones above. The striations on the top and bottom of the crystal remain, but they have been “squashed” lengthwise so that the crystal is no longer quite as long, but much thicker.
There are pros and cons to growing them from a saturated oxalic acid solution. The good news is that there was significantly less mold in the highly acidic solution. The bad news is that oxalic acid crystals also formed simultaneously, and the two types of crystals frequently got stuck together:
 The transparent plates are crystals of oxalic acid, grown into the green potassium ferrioxalate crystals.
The transparent plates are crystals of oxalic acid, grown into the green potassium ferrioxalate crystals.
In short, if you want long crystals, grow them from a less acidic solution. If you want thicker and stockier ones, grow them from a more acidic solution.
That’s all I have to say about impurities.
Finally, I’d like to mention that it’s also possible to grow crystals from other ferrioxalate compounds, like sodium ferrioxalate and lithium ferrioxalate. Substituting potassium carbonate, the preparation and growing process is exactly the same.
They have different crystal structures, and I plan to try them out in the future.
I hope you enjoyed the article. Feel free to drop a comment if you’d like to ask me anything.
Also, if you’d like to read more articles like this one, check out my post on how to grow stunning crystal clusters from fertilizer.
As always, happy growing.
will the black rust from blacksmithing work?
I’m not sure of the composition of your black rust – but black iron oxide won’t work – you need red iron oxide.
Will this crystal starts melting when we heat it in water?
This crystal will dissolve in any water – heated or not.
Thank you for your comprehensive instruction on growing Potassium Ferrioxalate crystal. I find it fascinating to create the solution by three compounds instead of simply buying one compound. I am planning to make this crystal. I am going to purchase from Amazon the iron oxide Fe3O4 (from AC Alpha Chemicals), potassium carbonate (from Essencea) and oxalic acid (Varomorus 99.6% pure). Are the manufacturers of these products acceptable?
Another question: You mentioned that “the surface of crystals exposed to bright light will turn duller, and eventually white.” What will happen if the crystal is encapsulated in epoxy inside a plastic transparent container — Will the epoxy prevent the crystal from de-coloring? Will the heat of the epoxy curing process create a shiny surface on the crystal?
Thank you for your attention.
Kim Wong
When are you going to turn this into a video on your channel? I’m waiting for more content! You only have one YouTube short.
There are 6 vids there right now. Still working on this one 🙂
Can I use FeCl3 instead of Fe2O3?
Yeah, many tutorials online prefer this method
Twice the mole of Fe2O3 right?
Keep it up and I hope you will make more and post them on YouTube again.
Hi Chase,
Thank you for these turorials, I have learned a lot more about crystals than what I knew previously. I will br trying a couple of the recipes that you have listed here.
My big question for you, is have you ever tried growing gypsum / selenite crystals? I have never found any directions for doing this, but surely it must be feasable in a reasonable amount of time. I have tried only using water, and failed. Maybe adding Oxalic acid would work? Please let me know your thoughts.
Gypsum (calcium sulfate) is rather insoluble in water, which makes it very hard to grow crystals with. It is slightly more soluble in salt solution, so you might want to give it a try.
Here’s a Reddit post I made about it a while ago: https://www.reddit.com/r/crystalgrowing/comments/wvpmqw/these_are_gypsum_calcium_sulfate_crystals_that_i/
Never managed to grow any bigger ones though.
Do you know what chemical potassium ferrioxalate turns into when reduced by light? Ferric or ferrous oxalate? Also, how long does it take for this chemical to oxidize back into potassium ferrioxalate? Thanks, very helpful!
Ferrous oxalate. Not sure how long it takes to turn back, but solutions definitely turn much greener after sitting in the dark for a few days.
Hey! Thanks for these tutorials.
For some reason it seems like my iron oxide isn’t dissolving properly. After a couple of hours, the liquid levels go down to about where yours are in the pics – but the colour is still very reddish brown. Is there something I might be doing wrong (I’m definitely using the prescribed 45g of oxalic acid, 200ml of warm water, and 6g of iron oxide)?
Hey Jay, a few people have this problem too. It’s possible that some iron oxide has indeed reacted. Try filtering out the solution anyway, and see if the solution is clear or brownish. If it’s brownish, then you can already proceed to the next step.
If it’s clear, that means no iron oxide has dissolved at all. If so, check if you are using red iron (III) oxide. There are other varieties, which don’t work as well. Next, try adding a bit more oxalic acid and heating for longer. If it still doesn’t work, consider using other methods to produce iron (III) oxalate, like first reacting iron metal with oxalic acid to form iron(II) oxalate, and then oxidizing it with hydrogen peroxide. Good luck!
Hi Chase! Would you recommend covering these crystals with clear nail polish? (I’ve grown copper sulfate and potassium hexacyanoferrate crystals and covered both with clear nail polish to prevent them from drying out and make them safe to hold/touch).
Yup, you can certainly do so! But note that the main issue with potassium ferrioxalate crystals is that they break down with exposure to light. Therefore, unlike other crystals, nail polish might not protect the crystals from degradation.
Do you know of any way to use a seed crystal that doesn’t require fishing line? It would be nice to grow a larger crystal without having the small imperfection in its core.
Also, for someone working at home without access to a lab (no DI water handy) how would you recommend cleaning equipment?
There’s no simple way to grow a crystal that is well formed on all sides without a fishing line. You could also let the crystals continue growing on the bottom of the container, but the side facing the bottom will be flat. Alternatively, you can try to encrust a rock on a branch with a cluster of natural looking green crystals by placing multiple seed crystals with the orientation you wish.
Based on my experience, cleaning equipment isn’t terribly important ~ at least for the crystals shown in my guides. Just wash your containers with tap water, and wipe dry. If there are rust stains, soak it in a dilute solution of oxalic acid for a few hours and it should come off.
Making this compound was the very first exercise done by students taking first-year chemistry at my second university. (It’s also known as potassium trisoxalatoferrate.) The recipe involved precipitating ferrous oxalate from ferrous sulphate (?) and oxalic acid, followed by oxidation with hydrogen peroxide and addition of potassium oxalate, if I recall correctly. It was a nice exercise, with pretty colours and a tangible “look at what I made!” result. It also let the lab TAs get an early idea of who was and was not able to follow a straightforward recipe.
Inevitably, some students dropped out after only a week or two, leaving their beakers of green solution behind in dark cupboards in the labs. At one point, I was helping the lab manager to do some cleanup months later, and found one of those beakers after the crystals had been growing for months. There were some truly lovely crystals there.
One catch with preparing ferric oxide from steel wool is that the metal may be impure. A couple of years ago, I was trying to make some ferric oxide, and used bleach (sodium hypochlorite) as an oxidizer with steel wool. I was surprised to see the solution turning purple. It turned out that I was getting permanganate in solution, from manganese in the steel wool.
My university also had a lab where we were supposed to grow sodium chloride crystals, and then view them under the microscope. I was so excited for it, until it got cancelled due to the Covid lockdown. Sigh.
I know right. Crystals grow best when they are undisturbed. Sometimes the best specimens are those that you totally forget about, and then come back several months later to see absolutely wonderful formations. Of course, that’s provided that the solution hasn’t completely dried up, in which case the entire crystal would be coated with a dry crust.
A decade or so ago, I decided to try growing crystals of baking soda, just to see what I’d get. I prepared a saturated solution: excess powder dissolved in warm water, let cool to room temperature so some precipitated out, filtered carefully into a tall glass. I put the glass on a top shelf where it would be undisturbed, with near-constant temperature, and left it covered, with only a tiny gap for evaporation… and completely forgot about it until months later. When I noticed it, it was completely dried out, and I had a *beautiful* object that looked like a glass sea urchin. Hundreds of long delicate spines growing from a nodule at the bottom of the glass. I don’t recall how I got it out — I’m guessing that I ran a tiny bit of water into the glass to weaken the structure’s attachment. It’s still on my mantelpiece, but all of the spines have broken short. It kept getting disturbed by people who didn’t take enough care with it.
Baking soda crystals. Very interesting. I’ve never seen any specimens of macroscopic sodium bicarbonate crystals before. Can you send me a photo of it?
Many other crystals like to grow needle-like clusters, from sodium acetate to calcium sulfate, but mine have never gotten sturdy enough to pull out of solution without damaging them.
Hi Chase!
Thanks a lot for your wonderful guide! As a beginner, it’s my very first time to make something in chemistry, and I’m just stuck at dissolving the rust. I followed the guide to prepare the solution and heat it on the hot plate with a temperature around 40-50 Celsius for 18 hours, but the color is still reddish-brown and the solid is not seem to be resolved. I’ve tried several times but none of them succeed. I’m wondering if I missed any steps?
Hello! That’s okay, I ran into the same problem too. Try heating at 80-90C. To speed things up, you can try adding excess oxalic acid straight away to the solution (since you need the excess to neutralize the potassium carbonate anyway). A more acidic solution will help. Use a stir bar if you have one.
If everything still doesn’t work, just wait for the solution to cool, and filter it into a new container. Chances are at least *some* rust will have dissolved, so the solution should be brownish. This is a good sign, and it means you can proceed to the next part of the reaction. Of course, you’ll end up with less product, but that’s fine. As for the leftover rust powder, you can repeat the procedure to dissolve more of it.
Don’t be discouraged. Chemistry is like that sometimes. The results will be worth it. Good luck!
Hi Chase,
I’m trying to do this one at home and for the Iron Oxide I took some iron bars from an abandoned building site near my house and oxidized them, but a white dust (zinc hydroxide) started forming on it as well. Will it affect the solution or the crystals in any way? And also, will a UV-resistant coating prevent the crystals from dissolving?
Thanks in advance, love your tutorials!
It shouldn’t be a problem. When you dissolve the iron oxide in oxalic acid, the zinc hydroxide will be converted into zinc oxalate, which is very insoluble in water. You can just filter it out before adding potassium carbonate. Do you mean whether the UV-resistant coating will stop the crystals from turning white? I’m guessing it will help, since it absorbs more of the high frequency electromagnetic spectrum. But I don’t think that will prevent decomposition entirely, because visible light also contributes at a slower rate.
Hi Chase. Can you help me convert from solid to liquid? FeSO4.7H2O -> FeSO4(liquid). And sorry my english is not good.
What do you mean by conversion? Also, do you mean from solid(s) to aqueous(aq)? FeSO4.7H2O has a molar mass of 278 g/mol, while FeSO4 has a molar mass of 152 g/mol. This means that if you are using FeSO4.7H2O for a reaction, you need to add 83% more by mass compared to if you just used FeSO4.
Wow. This would be the coolest component for board game pieces. Are these safe to handle? How hard are they?
Well, they are a mild irritant. It’s fine if you handle it occasionally and wash your hands afterwards. But holding it frequently isn’t recommended.
Plus, potassium ferrioxalate is sensitive to light, so it’s not a good idea for board game pieces. You don’t want them turning dull after a few games. Maybe use copper sulfate (blue) or alum (transparent) crystals instead, after sealing them in resin? I have written a guide on them as well.
This compound is fairly brittle, and not particularly hard, unfortunately. It’s probably about on a par with salt crystals.
can you grow crystals from aluminum oxide as well?
Aluminum oxide is insoluble in water, so no. However, it can be reacted with other chemicals to form compounds that do form crystals, such as potassium alum and sodium magnesium oxalatoaluminate.
Hi Chase, thanks for the guide. Is it possible to put a resin or clear lacquer around the crystal to set in jewelry? Or will the exposure to light and heat degrade the crystals?
Unfortunately, it’s not possible for this particular crystal as light exposure is still an issue. It might work for other crystals like alum and copper sulfate.
Super cool!! I was wondering, is it possible to grow some of these types of crystals like, on stuff? I’ve been wanting to try growing crystals on skulls/ceramics for a while now – would the solution compromise the integrity of the material I was trying to grow on? And would there be any way to control where the crystals would grow/not grow? (If these questions are too much of a pain to answer, that’s fair lmao, just figured I’d check) Thank you!
That’s actually a very good question. It’s possible, and in fact, many people do it. Most of the crystals shown on this website are not very acidic, so they will not compromise the integrity of the object. Generally, the best way to get crystals to stick to the object is to make sure that the surfaces you want them to grow on are rough, while the other surfaces are smooth. Also, try to get the solution to be supersaturated.
The most common compound people use to crystallize stuff is MAP (a type of non-toxic, common fertilizer). I haven’t written a guide on that yet, you can check it out online. Other alternatives are potassium alum, borax and copper sulfate.
Good luck!
Hi Chase,
Once more, what a beautiful guide! Thanks a lot!
Total beginner here. You are always wearing gloves when holding the crystal. Does this mean you can’t touch it with bare hands? Thought it would be ok after you’ve got the crystal.
Thanks again!
Thanks! It’s actually fine to touch the crystal, just wash your hands afterwards. I wore gloves because it’s generally a better practice, and I also because I didn’t want to get the crystal dirty.
It’s rather water-soluble, so handling it with bare skin carries some risk of smudging a shiny crystal surface by contact with perspiration.